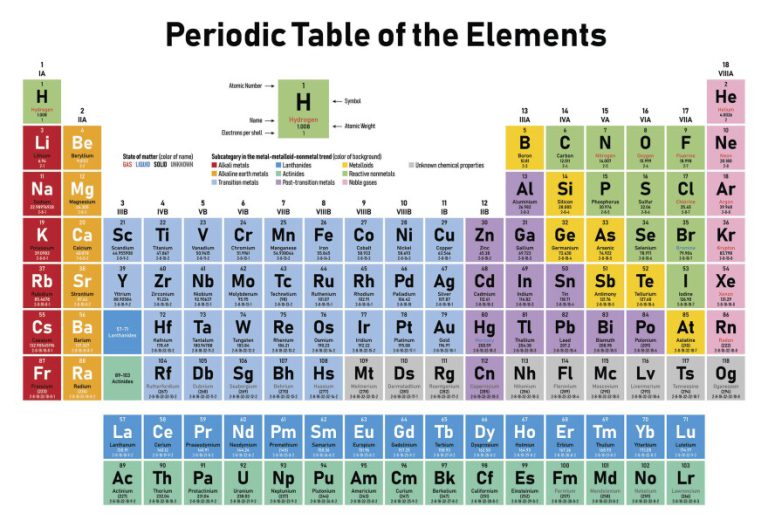
Mandeleev law says that the properties of elements are the functions of their atomic mass.Henry Moseley arranged the elements according to the increasing atomic number.Mendeleev arranged the elements according to the increasing atomic mass.This law is exactly similar to the Mendeleev law, but the only difference is Modern Periodic Law was given by Henry Moseley in 1913. The properties of the elements are the Periodic function of their ATOMIC NUMBERS. Yes, all the elements in modern periodic table are arranged on the basis of their ATOMIC NUMBER. We have seen in previous section that Dobereiner’s law, Newland’s law as well as Mendeleev law were based on the arrangements of elements according to their increasing atomic masses.īut the only difference in modern periodic law is I have also discussed the merits and demerits of the Mendeleev periodic table. I know this seems a little difficult for you, so you can refer to this detailed guide on “ Mendeleev periodic table” where I have explained all these things with proper images for your better understanding. So there were some empty blocks seen on the Mendeleev periodic table. 
Now look, while arranging the elements according to atomic masses, Mendeleev found that the properties of few elements were not matching with any of the previous ones, so he didn’t place such elements in that particular column. 
Why are some blocks empty in the above Mendeleev periodic table? That’s why Mendeleev placed those elements in the same block. The short answer: Those elements show similarly in their properties. Have you noticed that many blocks have more than one element in Mendeleev periodic table? Why? There are a total 8 vertical columns in the Mendeleev periodic table, which are known as groups.Īnd there are 7 horizontal rows, which are known as periods. Write the valence electron configuration of each element by first indicating the filled inner shells using the symbol for the nearest preceding noble gas and then listing the principal quantum number of its valence shell, its valence orbitals, and the number of valence electrons in each orbital as superscripts.Ī The group 2 elements are in the s block of the periodic table, and as group 2 elements, they all have two valence electrons.This table is known as the Mendeleev periodic table of elements.Locate the nearest noble gas preceding each element and identify the principal quantum number of the valence shell of each element. Identify the block in the periodic table to which the group 2 elements belong.Use the periodic table to predict the valence electron configuration of all the elements of group 2 (beryllium, magnesium, calcium, strontium, barium, and radium).Īsked for: valence electron configurations For elements after No, the electron configurations are tentative. The electron configurations of the elements indicated in blue are also anomalous, but the reasons for the observed configurations are more complex. The electron configurations of elements indicated in red are exceptions due to the added stability associated with half-filled and filled subshells. \): Electron Configurations of the Elements.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
